21 CFR Part 11 Compliant Software
INSTECH SYSTEMS offers software solutions that are fully compliant with the stringent regulations of 21 CFR Part 11 This regulation is set by the FDA which ensures that electronic records and signatures are reliable secure and equivalent to paper records Our software is specifically designed for sectors such as pharmaceuticals biotechnology and healthcare where regulatory compliance is of utmost importance,
INSTECH SYSTEMS you get a solution that supports the integrity security and auditability of your digital data and is fully compliant with Part 11 Our software offers robust features such as secure login protocols electronic signatures and audit trails to track all user activities which helps prevent unauthorized access and ensures that all records are legally valid and traceable ,We understand the importance of regulatory compliance and our solutions are built with the latest technology to seamlessly integrate with your existing systems Trust INSTECH SYSTEMS and get secure reliable and compliant software that matches the highest industry standards
Instech JM.S Development and Function:
Instech JM.S includes a set of functions for responding to the requirements specified in FDA 21 CFR Part 11. The standard is intended to provide a solution for securely handling electronic records and electronic signatures in industrial applications.

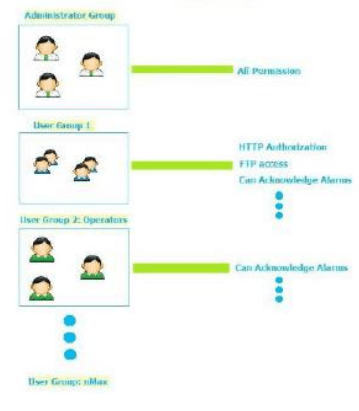
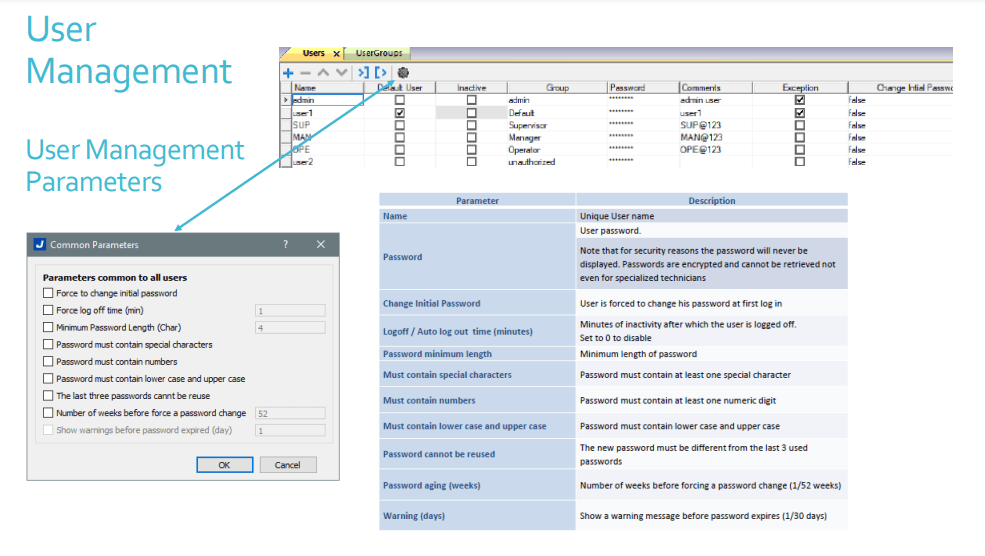
User Management Security
- Instech JM.S provides customizable user group dynamics
- You can restrict access to various widgets and operations by configuring users, user groups and assigning specific authorizations to each group
- Max User Groups = 50
- Max Users = 500
- Each user must be member of one and only one group. Each group has specific authorizations and permissions.
- By organizing permissions and groups you can define the security options of a project
Benefits of 21 CFR Part 11 Compliant Software
- Legal Approval – Electronic records and signatures are considered legally equivalent to paper records which are valid for regulatory purposes
- Better Security – Features like encryption and secure login are provided to keep sensitive data safe
- Audit Trails – Every activity is tracked, which maintains transparency and makes audits easier
- Easy Compliance – This software complies with FDA regulations in an automatic way which makes compliance very easy
- Fast Work – Automates workflows to reduce paperwork and speed up work
- Risk reduction: The risk of fines and legal issues arising due to non-compliance is significantly reduced
- Grows with business – As your business grows, this software adapts according to your needs.
- Regular Updates – The software is always updated with the latest regulations
Using 21 CFR Part 11 compliant software secures your data makes work efficient and reduces compliance-related risks
What is the purpose of 21 CFR Part 11?
The primary purpose of 21 CFR Part 11 is to define the requirements for the use of electronic records and electronic signatures in FDA-regulated industries. It ensures that electronic records are secure, reliable, and legally binding, just like traditional paper records and handwritten signatures.
Which industries are affected by 21 CFR Part 11?
Industries that are subject to FDA regulations, such as pharmaceuticals, biotechnology, medical devices, food manufacturing, and clinical research, are typically affected by 21 CFR Part 11.
What are electronic records under 21 CFR Part 11?
Electronic records are any information created, modified, maintained, archived, retrieved, or transmitted electronically, including text files, databases, spreadsheets, and other digital formats.
What are electronic signatures under 21 CFR Part 11?
Electronic signatures are digital representations of handwritten signatures, which can be used to sign electronic records. These signatures are used to authenticate and link an individual to the electronic record.
What are the consequences of non-compliance with 21 CFR Part 11?
Non-compliance with 21 CFR Part 11 can result in regulatory actions, including warning letters, fines, product recalls, and legal consequences. It’s essential for regulated industries to ensure compliance to avoid these risks.
How can organizations achieve compliance with 21 CFR Part 11?
Achieving compliance involves implementing appropriate policies, procedures, and technologies to ensure the secure and reliable use of electronic records and signatures. This typically includes system validation, data encryption, access controls, and the establishment of audit trails.
